


Background:
The integration and networking of medical equipment has become an indispensable component of modern operating theatres. At present, the market is characterized by closed solutions, which are regulatorily approved as monolithic settings. The aim of the project is therefore to develop methods that support the development of openly integrated medical devices as well as the approval process by means of a test environment. The project aims to ease the access of SMEs to the market with innovative technologies.
Material & Methods
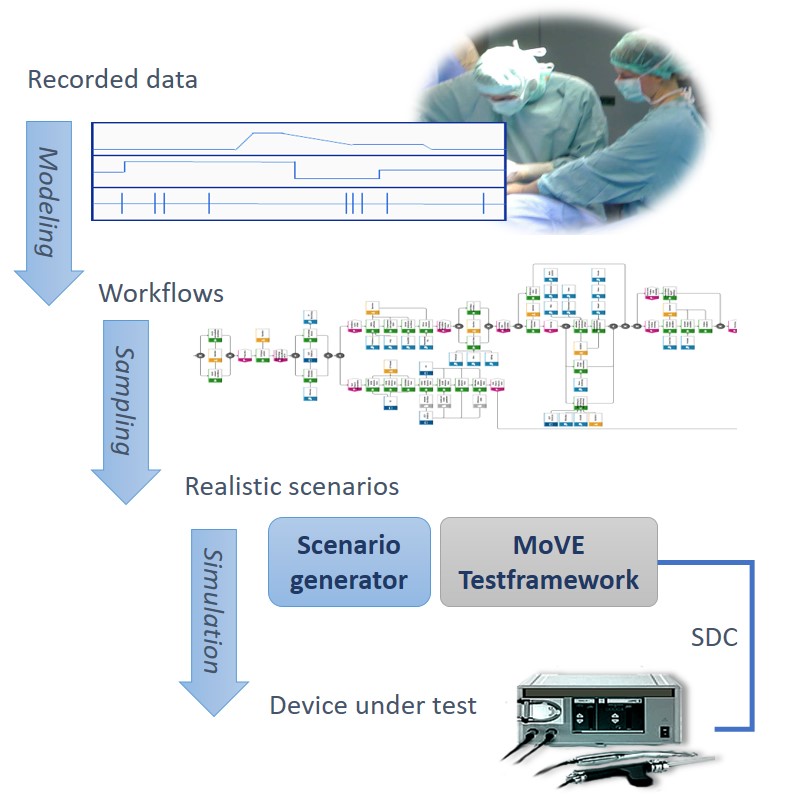
A simulation environment, including communication infra-structure, simulated medical devices and test scenarios, is being developed. The test platform will verify the networking of medical devices and software components and validate the communication regarding conformity with the communication standards, appropriate message and communication patterns, and timing aspects.
Realistic, scenario-based simulations of an OR setup and its communication are implemented by a simulation engine and emulators of medical devices. Based on an integration with the IEEE 11073 SDC standards family, manufacturers can test their products against the virtual infrastructure early on in the development process.
In the frame of the project, ICCAS is responsible for the automated generation of realistic test scenarios from recordings of real interventions. Methods are developed that use already well-developed stochastic workflow modeling approaches to sample possible intervention courses and orchestrate the states and parameters of the emulated devices based on the estimated surgeon’s behavior. To that end, a simulation engine is implemented, which reads formal descriptions of user and device behavior. A network of sampling components of various types, including Hidden Markov Models, Random Forests, and empirical distributions, is set up and is frequently updated upon the run of a scenario to generate realistic measurements, device parameter configurations, and remote procedure calls for the device under test.
Results
In December 2019, we were able to present the results of the project in presentations and several demo stations. In a first step, the basic interoperability of medical devices using the IEEE11073-SDC standard family was checked. In a second step, complete surgical interventions with a variety of real and simulated medical devices could be performed using a complex simulation framework. In particular, the behavior of the device under test and the network interface can be checked. In the last demo station, the individual components were brought together, and the corresponding result reports were generated based on the results of the test runs. The feedback from the 50 participants in the final presentation on the results shown was consistently positive.

Um dir ein optimales Erlebnis zu bieten, verwenden wir Technologien wie Cookies, um Geräteinformationen zu speichern und/oder darauf zuzugreifen. Wenn du diesen Technologien zustimmst, können wir Daten wie das Surfverhalten oder eindeutige IDs auf dieser Website verarbeiten. Wenn du deine Zustimmung nicht erteilst oder zurückziehst, können bestimmte Merkmale und Funktionen beeinträchtigt werden.