Sponsors:
The project will investigate whether the expression of a specific gene in gastric tumors can be determined solely from tissue sections after routine staining and using advanced deep learning techniques. This could optimize and potentially replace current methods, which are costly and time-consuming. For this purpose, an extensive data set from an already completed multicenter study and complex neural networks will be used. The results could lead to faster and more cost-effective diagnostics and personalization of treatment for patients with gastric cancer. The models and results will be made publicly available to support further research and possible integration into clinical cancer treatment.
Sponsors: European Commission,
The EU consortium “CERTAINTY – A Cellular Immunotherapy Virtual Twin for Personalized Cancer Treatment” has launched a 4.5-year project to implement virtual twin technology for cancer therapy personalization. Immunotherapies have gained traction in oncology, involving comprehensive data collection to guide diagnosis, treatment, and monitoring. ICCAS aims to harness these data through virtual twins that simulate disease prognosis and treatment outcomes. The project develops technological infrastructure for virtual twins, multimodal data evaluation, personalized predictions, and context-specific user interfaces to support clinical decision-making.
CERTAINTY integrates various analysis methods and prediction models into a comprehensive, multimodal representation of patients, enhancing personalized simulations and clinical decisions. A modular virtual twin for cancer treatment is being created, initially focusing on CAR T-cell therapies and multiple myeloma. The twin will reflect the unique pathophysiology of each patient, updating continuously during treatment, emphasizing molecular patterns, big data processing, machine learning, in vitro models, and mechanistic models. The project prioritizes secure interfaces for data access while considering socio-economic factors and future applications.
Sponsors: KARL STORZ SE & Co. KG,
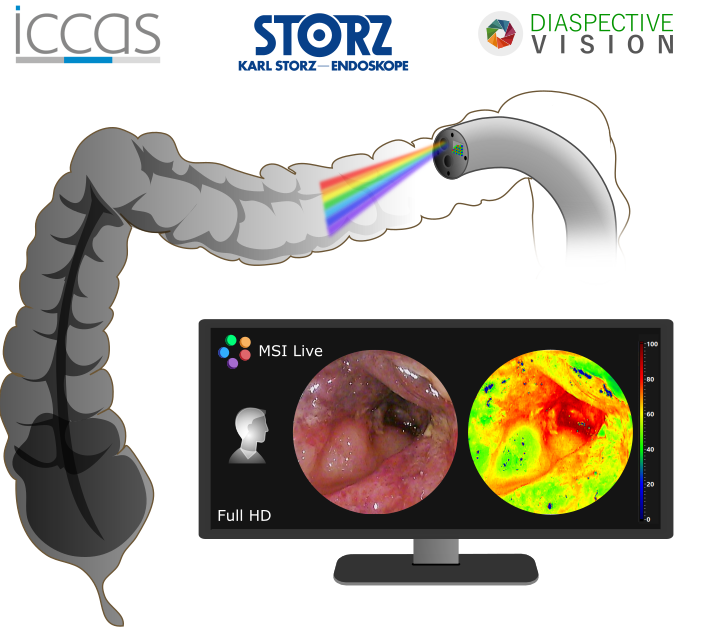
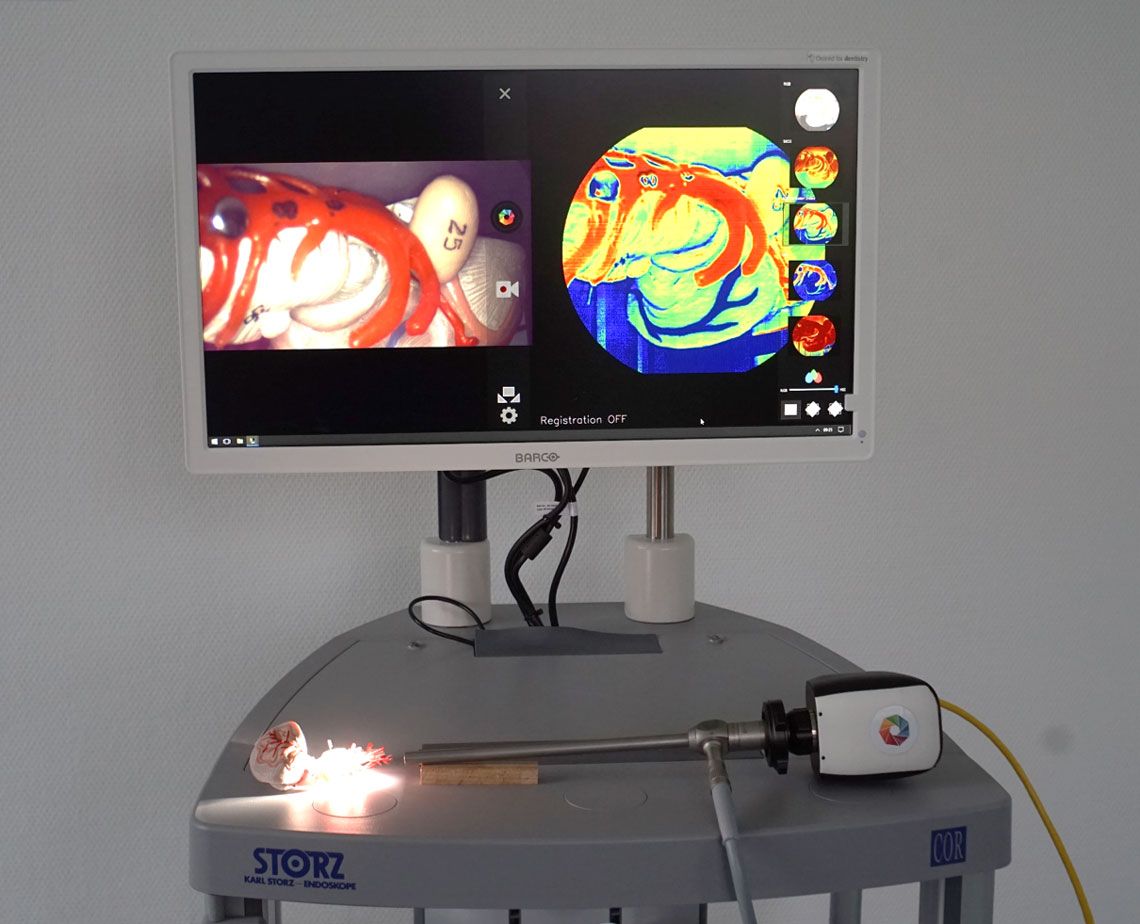
Multi- and hyperspectral imaging (MSI and HSI) can support the intraoperative assessment of tissue perfusion, identification of tissue structures and tumor tissue. The aim of this project is to acquire and analyze spectral data with different MSI and HSI systems in a (pre-)clinical environment. Potential clinical fields of application and limits of the technology will be evaluated. Furthermore, procedures for the automatic classification and visualization of tissue using spectral image data are developed and implemented. The resulting information will be available to surgeons intraoperatively using augmented reality methods.
Sponsors: Federal Ministry of Research, Technology and Space,
The DaDriv-StAC project aims to develop a digital platform that enables data-driven decision support in the aftercare of stroke patients. The focus is on researching and applying the innovative concept of virtual medical twins. These will integrate and analyse complex health data from different sectors – such as acute hospitals, rehabilitation facilities and the outpatient sector – and compare it with standardized and individual treatment paths. Artificial intelligence (AI) will be used to derive treatment recommendations that support personalized and guideline-based treatment throughout the entire course of care.
As stroke aftercare is particularly challenging due to individual risk factors, secondary diseases and long-term restrictions such as paralysis or speech disorders, DaDriv-StAC aims to improve cross-sector networking and continuous therapy monitoring. To date, there has been a lack of corresponding technological platforms that dynamically update, network and evaluate health data in a meaningful way for those treating patients. The project is the first to implement the concept of a dynamic virtual twin in stroke aftercare, which continuously adapts to the course of treatment and thus enables data-based feedback to practitioners and patients.
Leipzig University Medicine (UML) is playing a central role in this. Together with Actimi GmbH, it is developing the platform and conducting a clinical feasibility study to evaluate acceptance, effectiveness and practicability. In addition, the data collected will be used for medical quality assurance and process optimization.
The technological core of the project is the creation of a new data model that integrates medical, clinical and subjective patient data (e.g. PROMs/PREMs). Based on this, new analysis and decision-making methods will be developed that enable a more precise assessment of risk factors and the comparison of individual progressions with optimal treatment paths.
The project thus addresses key challenges facing the healthcare system: it aims to close information gaps, improve treatment quality and contribute to more efficient, personalized care in the long term. The results will be both scientifically published and prepared for later commercial scaling.
DaDriv-StAC meets the objectives of the funding line for the development of data-driven decision-making and support systems in the healthcare sector and makes an important contribution to digitalization and innovation in medical care.
Sponsors: ZIM – Central Innovation Programe for Small and Medium-sized Enterprises,
Despite major scientific advances, neonatal sepsis and premature infant apnoea remain key challenges in neonatal intensive care medicine. Late-onset sepsis is blood poisoning that occurs at the earliest 72 hours after birth and is usually caused by hospital germs. Apneas (breathing stops) occur in almost all premature babies and often lead to critical drops in oxygen saturation (SpO2}. Although current monitoring makes it possible to detect sepsis and apnea, it requires numerous contact electrodes, which can cause skin damage in immature premature babies. In addition, apnea is often only detected through its effects (drop in SpO2). The aim of the project is to develop a contactless measurement method for recording respiratory movement and body temperature for sepsis and apnea detection, based on the time-of-flight measurement of electromagnetic waves and optical temperature measurement, in order to reduce the number of wired sensors.
Sponsors: ZIM – Central Innovation Programe for Small and Medium-sized Enterprises,
As part of the FaszioTrain project, a realistic training environment for fasciotomies on the lower leg is being developed in collaboration with Phacon GmbH. To this end, various types of tissue are being produced using rapid prototyping methods and integrated into a model that replicates the physical properties, colours and textures of real anatomical structures. The model will consist of several anatomical structures that realistically reproduce the real anatomy of the lower leg, including the various compartments, nerves and vessels. To enable the realistic simulation of fasciotomy, the model will simulate the swelling of blood congestion in the tissue and the resulting pressure on the nerve tracts. Users should learn how to open the tissue properly, work their way to the release point and achieve the best possible relief.
Sponsors: Federal Ministry for Digital and Transport (BMDV), TÜV Rheinland Consulting GmbH,
The project aims to evaluate the performance of modern 5G campus networks for XR-based medical applications. The objective to examine collaborative XR-based telemedicine from a technological and user-centric standpoint, to assess its effectiveness, and suitability for clinical use. To achieve this, a technology demonstrator will be designed and developed to enable testing of an XR-telemedicine application over 5G campus networks. The resulting application will be evaluated by various end-users from relevant clinical fields. Anticipated challenges are synchronization of data coupled with low latency for the targeted cloud and edge-cloud use cases. The partnership between LeFx and ICCAS combines cutting-edge XR visualization with profound experience in campus networks based on 5G technologies for medical applications. Collaboration with Telefonica further enables the employment of modern managed service networks, which can be used in addition to ICCAS’s own 5G campus network. These networks will be managed and modified for the intended XR applications. Medical use cases for the technological demonstrator will be developed and tested by a consortium of clinical experts from Helios Parkkankenhaus Leipzig and the Universitätsklinik Leipzig. The UML contributes its extensive medical expertise through collaboration with specialized clinics, its own expertise with the development of AR/XR applications, as well as experience in establishing and operating 5G campus networks.
Sponsors:
The focus of the project is on the interoperability of medical devices and high-bit-rate wireless data transmission using 5G, among other technologies. The goal of ICCAS in the project.Open5GPaceMaker is to integrate Time Sensitive Networks (TSN) into an existing medical device in a hospital environment. To achieve this goal, the clinical requirements for signal transmission in medical devices, as well as current technical solutions from medical device manufacturers and communication technology vendors.
For Open5GPaceMaker, a demonstrator will be produced that allows real-time wireless transmission of its control commands within an operating room using TSN over 5G. Through an existing and long-standing collaboration with the University Hospital Leipzig, realistic tests of the developed demonstrators are performed. On the level of communications engineering, ICCAS works closely with project partners and contributes experience from the field of medical applications. In cooperation with the University Hospital Leipzig, the clinical requirements for TSN over 5G in a hospital environment will be determined and made available to the project partners. The results will be implemented and tested in the LivingLab
Sponsors: European Union, Sächsische Aufbaubank – SAB, Saxon State Ministry for Science and Art (SMWK),
The aim of the project is to establish defined processes for the fabrication of patient-specific and application-specific soft end effectors.
… (more informations)
Sponsors: ZIM – Central Innovation Programe for Small and Medium-sized Enterprises, Federal Ministry for Economic Affairs and Climate Action,
The SurgiTrace project aims to develop an intelligent, AI-supported system for effective resource planning during surgical procedures. … (more informations)
Sponsors: Federal Ministry of Research, Technology and Space,
The SUSTAINET-guarDian project is developing secure, resilient and sustainable mobile communications infrastructures based on a further development of today’s 5G technologies towards a service-based architecture. It deliberately avoids a completely new 6G generation. The focus is on a robust security architecture, automated network control and the targeted use of artificial intelligence (AI) to make networks efficient, reliable and scalable.
Important goals are the standardization of authentication and encryption, protection across untrusted network segments and the protection of AI over its entire life cycle. Cyber resilience is strengthened by modern measures such as homomorphic encryption, quantum key exchange and trustworthy cloud usage.
Another focus is on sustainability and energy efficiency. This includes distributed learning, efficient algorithms and methods for reducing energy consumption. The solutions developed will be tested and optimized using practical demonstrators in the areas of smart grid and medical applications.
Sponsors: TÜV Rheinland Consulting GmbH,
The TeleNoma project aims to develop an improved communications infrastructure for mobile medical teams and facilities in rural and underserved areas. The core of the project is the use of nomadic radio networks and the integration of 5G technologies in an open platform to provide a cost-effective and scalable solution for telemedicine applications. The project aims to develop a hardware-software kit that establishes a temporary nomadic 5G radio network and realises a seamless connection of medical devices and applications for mobile medical teams in an easy-to-use solution. By combining nomadic radio networks and telemedicine, a new overall technical concept is to be developed that can bring medicine at the level of a maximum care provider to underserved areas. The availability of a suitable communication and medical IT infrastructure should enable efficient medical services such as diagnoses, treatments, vaccinations, consultations and preventive medical check-ups as well as the ad-hoc deployment of medical teams in special or crisis situations.
Sponsors: Saxony, European Union,
Due to rapid advances in medical and pharmacological research, physicians today have increasingly effective options for the treatment of cancer. However, as the appropriate drugs are increasingly tailored to the specific characteristics of the patient and disease, new challenges arise in taking into account the complex diagnostic data and available therapeutic information (e.g. clinical trials). Since 2020, the Innovation Center Computer Assisted Surgery (ICCAS) at Leipzig University Medical Center, together with the Clinic for Hematology, Cell Therapy and Hemostaseology at Leipzig University Hospital, has been developing the IT platform “KAIT” – a comprehensive system for the analysis of medical information, which is intended to support the process of therapy decision-making in the clinical picture of multiple myeloma in the long term. The aim is to provide the treating physicians with all the latest information on the available therapeutic options so that every decision can be made on the basis of the latest findings. This will make an important contribution to the treatment of myeloma patients in Germany, regardless of where they receive medical care. Within the VOLTA project, this new and innovative form of clinical assistance will be further developed and extensively tested and optimized with clinical experts. These measures will ensure that “KAIT” can make a reliable and safe contribution to everyday clinical practice in the long term.
Sponsors: European Civil Protection and Humanitarian Aid Operations (DG ECHO),
The European medical response system is comprised of first responder units that are operating quickly and lightly. On occurring disasters (e.g., earthquakes, tsunamis, floods, etc.), these Emergency Medical Teams (EMT) are deployed on disaster relief missions to support the local medical system and avert humanitarian crises. As part of the “RescEU EMT” project, the European medical response system is being expanded to include modular, flexible units and special cells.
The “EMT Operating System” (EOS) is a field hospital information system, which is tailored to the requirements of EMT on disaster relief missions. Its idea was created and designed during the EUMFH-Project. The system supports the whole patient treatment process from triage to discharge and is highly configurable to adapt to the needs of the EMT. Despite EOS being primarily designed as an electronic patient record, it also includes essential functions for EMT mission and field hospital management. Besides patient management and treatment documentation, EOS enables quick department configuration, visualization of important hospital key performance indicators (patient admissions, triage category count, department workload, etc.) and reporting functionalities (e.g. to local government or WHO). Thus, EOS plays an essential role in monitoring and assessing the current situation and performance on a strategic and tactical level.
EOS provides highly customizable functionalities. They can be adjusted to the specific frameworks of different EMT entities or other requirements by specialized teams, e.g. Burn Assessment Teams. Generally speaking, EOS includes digital documentation and management of the usual processes within an EMT. However, detailed characteristics can differ.
EOS relies heavily on structured data entry and storage (in contrast to free texts). This ensures high information quality and supports fast and easy data input as well as automatic information aggregation in databases. The latter benefits the reporting obligation and allows for comparison between different missions or EMT installations.
Sponsors: avateramedical GmbH,
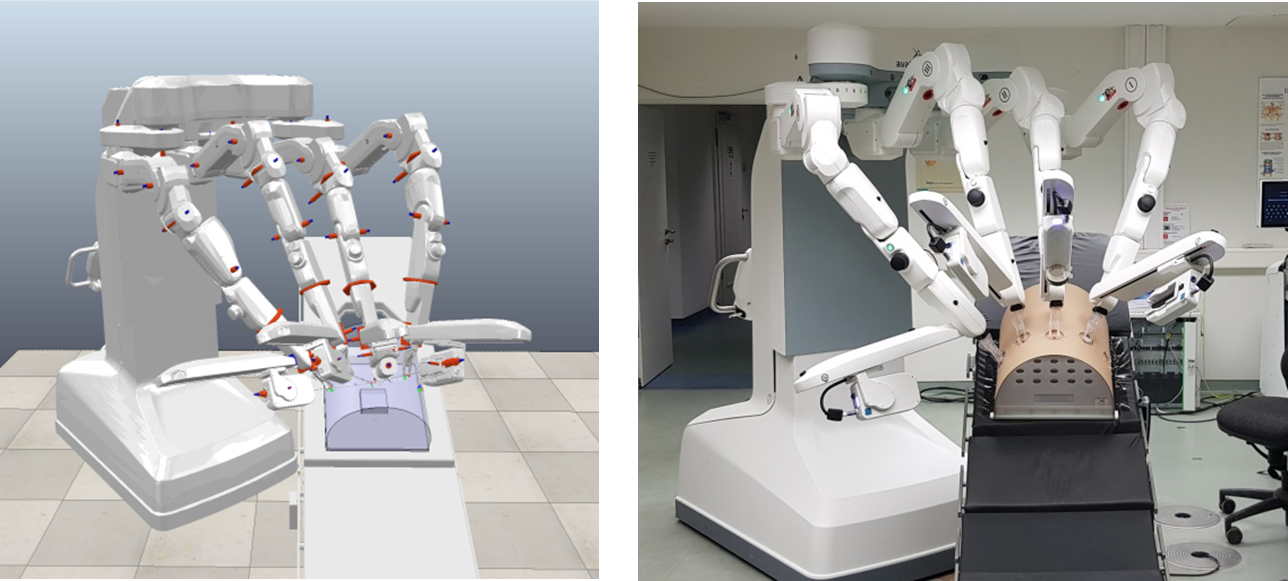
Under commission by the avatera medical GmbH (https://www.avatera.eu/home), ICCAS is investigating the possibilities to optimize the positioning principles of a novel robotic system for laparoscopic interventions. In cooperation with the urology department of the University of Leipzig Medical Center, the requirements for the pre-positioning of the system at the OR-table were defined and transferred into robotic workspace simulations.
The aim of this work is to provide an intuitive procedure for the docking process of the robot at the OR-table and an optimized positioning for the robot arms at the patient for the best possible workspace during the intervention. ICCAS developed a guidance manual for the side docking of the system and currently investigates the feasibility for radical prostatectomies and combined hysterectomies and Lymphadenectomies.
Sponsors: Federal Ministry of Education and Research (BMBF),
In practice, medical decision-making to determine suitable therapeutic approaches for an individual patient is subject to both a medical and a regulatory framework. Indication-specific guidelines, which indicate the treatment options approved (and thus applicable) in the respective health care system, form the formal basis for this. Although physicians can also act outside of these guidelines (so-called off-label use), they usually need good reasons and the approval of the respective payer. Furthermore, clinical trials are of course an essential factor in the evaluation of the best possible therapeutic option, although this aspect cannot be attributed to routine care and is dependent on numerous external factors (range of trials, suitability of the patient, randomization, etc.).
The fundamental question in any therapy-associated decision-making process is which available option offers the highest chance of success with equally low risk for the individual clinical picture of a patient. However, due to the heterogeneity of the individual and the disease, this consideration is very complex and requires the parallel consideration of multiple factors. The limitation of the therapeutic scope of action facilitating this circumstance does not solve the problem of the cognitive simulation of all possible implications in order to determine an appropriate optimum. Furthermore, there is the legitimate question which circumstance can be regarded as optimal at all, since the definition of success or failure may differ between therapist and patient. For example, a therapy could lead to tremendous clinical success, but equally come with the high risk of an invasive side effect. These sometimes life-changing decisions therefore require a high level of safety, education, and communication on an equal footing between all parties involved.
A technical possibility to provide assistance in the process of medical decision making are clinical decision support systems, which match specific characteristics of a patient (e.g. clinical findings) with a previously created knowledge base to quantitatively evaluate all available options. The VISION-CRE project therefore focuses on the establishment of a “Cognitive Reasoning Engine (CRE)”, which complements the guideline-based specification of possible therapy options with a novel evidence-based evaluation level. The overall goal is to extend the existing decision support tools by evaluating already collected and recorded empirical data of the sequence: (1) patient:in, (2)therapy and (3) resulting reaction in order to draw valuable conclusions for the treatment of patients.
Sponsors: European Union funding for Research and Innovation: Horizon 2020,
The GenoMed4All project is an EU-wide initiative for the establishment of a network-infrastructure to facilitate the exchange of clinical data in a federated learning framework. By integrating valuable clinical data up to multi-OMICS levels, the project aims to significantly improve quantitative analysis using machine learning (ML) and artificial intelligence (AI) methods. GenoMed4All focuses on a range of hematological diseases, which are becoming increasingly complex due to the particularly advanced utilization of precision diagnostics and personalized therapies.
In partnership with the Clinic and Polyclinic for Hematology, Cell Therapy and Hemostaseology at the University of Leipzig Medical Center, ICCAS is contributing to the mplementation of the federated data integration mechanisms as well as advanced data standardization based on HL7 FHIR.
External project site: https://genomed4all.eu
Sponsors: Federal Ministry of Education and Research (BMBF),
In the SaxoCell Systems project, ICCAS is developing mechanisms for secure tracking of necessary resources (analog and digital) in the context of ATMP development (e.g., findings, cells, pharmaceutical materials, etc.) on the basis of formal process models. For this purpose, the involved assets are first defined in the form of interoperable (HL7 FHIR) resources. Then, these digital images are instantiated within an IT platform based on a blockchain infrastructure and compared to an ideal process model with the principles of Good Manufacturing Practice (GMP). In the context of the SaxoCell overall project, the resulting supply chain management solution is intended to enable seamless traceability for ATMP manufacturing processes in order to enable superior and sustainable quality management in all areas of the intended platform.
External project page: https://saxocell.de

Sponsors: Federal Ministry of Education and Research (BMBF),
The vision of NFDI4DataScience (NFDI4DS) is to support all steps of the complex and interdisciplinary research data lifecycle, including collecting/creating, processing, analyzing, publishing, archiving, and reusing resources in Data Science and Artificial Intelligence.
The past years have seen a paradigm shift, with computational methods increasingly relying on data-driven and often deep learning-based approaches, leading to the establishment and ubiquity of Data Science as a discipline driven by advances in the field of Computer Science. Transparency, reproducibility and fairness have become crucial challenges for Data Science and Artificial Intelligence due to the complexity of contemporary Data Science methods, often relying on a combination of code, models and data used for training. NFDI4DS will promote fair and open research data infrastructures supporting all involved resources such as code, models, data, or publications through an integrated approach.
The overarching objective of NFDI4DS is the development, establishment, and sustainment of a national research data infrastructure for the Data Science and Artificial Intelligence community in Germany. This will also deliver benefits for a wider community requiring data analytics solutions, within the NFDI and beyond. The key idea is to work towards increasing the transparency, reproducibility and fairness of Data Science and Artificial Intelligence projects, by making all digital artifacts available, interlinking them, and offering innovative tools and services. Based on the reuse of these digital objects, this enables new and innovative research.
NFDI4DS intends to represent the Data Science and Artificial Intelligence community in academia, which is an interdisciplinary field rooted in Computer Science. We aim to reuse existing solutions and to collaborate closely with the other NFDI consortia and beyond. In the initial phase, NFDI4DS will focus on four Data Science intense application areas: language technology, biomedical sciences, information sciences and social sciences. The expertise available in NFDI4DS ensures that metadata standards are interoperable across domains and that new ways of dealing with digital objects arise.
Sponsors: Federal Ministry of Education and Research (BMBF),
As one of the recently extended German centers for Artificial Intelligence (AI), ScaDS.AI aims to close the gap between efficient use of large amounts of data in both industry and research and advanced AI methods. For this purpose, the research topics at ScaDS.AI range from foundational AI methodology up to the application of AI in key areas like engineering, environmental systems, industry, and biomedical research, cooperating with many local companies and scientific institutions. Furthermore, increasing the public trust in AI is taken into consideration by integrating ethical and societal perspectives and making research available through the service center and Living Lab.
To push forward AI in the life sciences, ScaDS.AI cooperates with ICCAS by sharing research expertise and computing resources, focusing primarily on model-based, personalized cancer treatments.
Sponsors:
The goal of ICCAS in the 5G-COMPASS project is to integrate current and future communication technology into medical (-technical) care. To achieve this goal, both medical device manufacturers and communication technology manufacturers must be supported in the development of their products. In the project, a test hardware will be developed that allows medical device manufacturers to easily test different communication channels (Wi-Fi, Li-Fi, 5G) and thus verify the performance of their products. On the other hand, realistic data is provided to the communication technology over different channels, thus enabling testing of the communication technology in a realistic environment. To this end, the company draws on its many years of expertise in modeling
surgical interventions and process automation. On the level of communications engineering, ICCAS is a user of the communications technology, works closely with the partners from communications engineering in the project and contributes requirements and experience from the field of medical applications. Selectively, it collaborates on the development of algorithms and configurations where specific adaptations for the field of medical applications are necessary. On the medical technology side, there is intensive collaboration with the partners KLS-Martin and SurgiTAIX. The results are implemented and tested in the LivingLab of ICCAS.
Sponsors: Federal Ministry for Digital and Transport (BMDV),
The Trimodal 5G (Tri5G) pioneer region stands for a 5G research and testing network in the northern region of Leipzig. The focus is on a wide variety of use cases with a focus on logistics and public supply, which are tailored to the requirements of industry and public services.
“Trimodal” stands for both the road, rail and air transport modes addressed in the project and the potential user groups of the technology to be researched from science, industry and public administration.
The Tri5G model region addresses the research, testing and standardization of 5G-based services in public mobile networks, tailored to the requirements of industry and public services for regional as well as supraregional logistics use cases. The goal is to create a 5G research and test network in the northern region of Leipzig, which is characterized by logistics traffic and automobile production. This network can be used to test and evaluate professional industrial as well as governmental 5G communication services in a public mobile radio infrastructure and under real conditions.
In Tri5G, a concept is to be developed to conceptually capture the future-oriented possibilities of 5G technologies mainly in the automotive industry, express logistics, value-added logistics and public transport in the vicinity of Leipzig Airport. In doing so, 5G use cases of different main users are to be combined in a unified network to avoid isolated solutions.
The 11 partners are investigating different use cases in the areas of Connected Driving, Automated Drones and Tracking, Monitoring, IoT & Maintenance.
The ICCAS task is to integrate a connected ambulance into a public mobile network and to explore the use of edge computing via 5G.
Sponsors: Saxony, Federal Ministry for Economic Affairs and Climate Action,
Digitization and interoperability for location and device-independent access to clinical data will be more firmly established in medicine. The greatest challenges at present are the lack of standardized, interoperable interfaces, the complexity of the clinical IT ecosystem, and the requirements posted by inevitable data protection and security measures.
In the MediNet project, the necessary interconnections between clinics, research and companies are being established. To this end, ICCAS runs a Transfer center to assist regional health care providers and companies with organizational and technical challenges when introducing novel products into the medical environment. Beyond that, ICCAS scientists are developing a technical platform that facilitates clinical studies and innovative treatment strategies with networked medical products. Based on established international standards and new technologies, the entire spectrum, ranging from mobile point of care devices to clinical information systems, can be addressed.
In cooperation with regional companies, the next generation of medical products will be integrated into clinical processes and digital infrastructures in the university hospital. This will also lay the foundation for new, more flexible solutions for location-independent, integrated medicine beyond the project period.

Sponsors: Federal Ministry of Education and Research (BMBF),
Overall project:
The 6G-Health project brings together the domains of communications engineering, medical engineering with medical and technical end users to enable precisely tailored technology development in the field of sixth-generation mobile communications (6G). This involves not only developing specific 6G technology components, but also identifying market entry barriers at an early stage and developing possible countermeasures. These include, above all, the aspects of licensing, operation and standardization. At the technical core are developments in the area of sensor integration in 6G, the development of technologies for enhanced network intelligence, and concepts and technologies for intelligent distribution of computing resources and efficient pre-processing of data at various levels of the infrastructure. From a medical perspective, representative applications from three areas will be addressed: The acquisition of biosignals directly at the patient and their transmission, the use and processing of data and information to improve a collaborative work environment, and applications of 6G in the field of Smart Hospital with the aim of recording and optimizing intraclinical processes.
Subproject – Leipzig University Medical Center:
Leipzig University Medical Center (UML) is developing ideas and concepts for the field of 6G research in medical technology applications. Technically, the main focus is on the investigation of application scenarios for the use of 6G technologies in clinical processes and their optimization. 6G can make a major contribution by providing a flexible, energy-efficient and high-performance infrastructure. The use of advanced sensor technology in clinics including the possibility of Joint Communication and Sensing (JCAS) as well as distributed processing intelligence opens up various advantages while significantly reducing power consumption. The medical applications addressed in the project are representative of different classes of medical applications. They are intended to highlight a wide range of future challenges and opportunities, which can be discussed with communication engineering experts via the 6G platform. In the project, the UML assumes the role of a mediator between the domains of medicine, communications engineering and medical engineering.Here, in the sense of a holistic understanding of the project, not only the technical applications are considered, but also the requirements for the future technologies through standardization and operation. To this end, the work is closely interlinked with players from the various domains and with experts from the areas of approval, operation, standardization and standardization.
Sponsors: Federal Ministry for Economic Affairs and Energy (BMWi),
The aim of the CortexMap project is the development of a novel navigated transcranial magnetic stimulation (nTMS) system for non-invasive pre- and post-surgical mapping of the motor cortex of the brain of patients with brain tumors. The new system is expected to offer an efficient use in neurosurgery and to be optimally integrated into the surgical workflow.
For this purpose, necessary hardware components as well as new software functionalities will be developed. An electromyography device with 8 or 16 electrodes for the measurement of motor evoked potentials (MEP) will enable faster and more precise examinations. Functionalities to automatically adjust the intensity of the stimulation and post-process the MEP will lead to accurate mapping of the motor cortex. New visualization and data analysis features will support the surgeons for the interpretation of the measurements too.
Therefore, the monitoring of patients before and after surgical treatment with this new non-invasive and simple measurement system, will become more efficient for the benefit of the patient.
Sponsors: ZIM – Central Innovation Programe for Small and Medium-sized Enterprises,
The SDC-VAS research project aims to develop a new ‘distributed alarm system’ for use in intensive care units based on the new IEEE 11073 SDC standard family. The primary goal is to reduce alarm fatigue and noise pollution in intensive care units.
The SDC family of standards is a new communication protocol that allows for communication between medical devices from different manufacturers. In doing so, these devices can provide data, status and services in an electronic network. This information can also be used by so-called value-added systems.
The distributed alarm system should receive the information from the various SDC-enabled devices and aggregate and evaluate it together with data from other sources such as the clinical information system (CIS) and IoT sensors and then forward said information to the appropriate nursing staff. In addition, we propose to explore alarm prediction possibilities using pattern recognition algorithms.
The project faces three core challenges: First of all, an integrator able to meaningfully link the data from the SDC interface, from the CIS and from various IoT sensors needs to be developed. Secondly, a meaningful methodology to select and inform a suitable employee needs to be established. Finally, the question to what extent the legal and normative regulations have to change in order to be able to use an SDC-based distributed alarm system safely needs to be answered.
The project, which is a cooperation between the company tetronik, the University of Leipzig represented by ICCAS and the HTWK Leipzig, has started in August 2022 and is funded by the ZIM program of the Federal Ministry of Economics and Climate Protection.
Sponsors: Federal Ministry of Education and Research (BMBF),
Funding measure „Recognizing and treating mental and neurological diseases Using the potential of medical technology for a better quality of life“
| Project title: | 3MP-FUS: Multimodality Multi purpose Multi plattform Focused Ultrasound – “Neuromodulation in rare neuropsychiatric disorders with focused ultrasound. ” |
| Principal investigator: |
Prof. Andreas Melzer |
| Project budget:: |
Funding amount/donation for the MF/UL: 5.2 Mio € (6.2 Mio € including fixed rate) |
| Project duration: | 01.04.2022 bis 31.03.2025 |
| Project partner: |
|
Project description
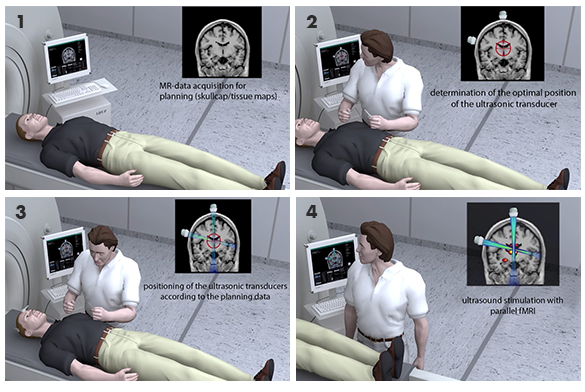
Clinically available MRI-guided FUS systems (MRgFUS/MRHiFU) are dedicated and approved only for specific indications (not to the diseases mentioned) and are approved only for specific MRI systems. In addition, they are permanently installed in one single MRI scanner. Flexible and cost-effective use of these FUS systems on other MRI scanners is currently not possible, in contrast to the planned 3MP-FUS system. One System to be used both under Ultrasound guidance and MRI guidance is not yet available.
The objective of this project is to demonstrate feasibility of such a multi use FUS System which we have developed for neuromodulation.
Neurological diseases are often accompanied by focal changes in the brain. Therapy options mainly aim to normalize the altered brain function, e.g. by neuromodulation. Deep brain stimulation (DBS) is in use for this purpose. DBS requires neurosurgical invasive intervention with the possibility for complications. An alternative would be non-invasive electromagnetic stimulation techniques, such as transcranial magnetic stimulation (TMS). However, these have a low spatial focus without reaching the relevant deep structures of the brain.
The BMBF funded project 3MP-FUS aims to optimize the neuromodulation adressing the two orphan diseases Dystonie and young onset Parkinson’s syndrome. Both benefit from invasive DBS but face the disadvantages of invasiveness. Dystonia (incidence of 20:100,000 (ORPHA:68363)) is characterized by spontaneous involuntary muscle movements. This disorder is usually treated with medication; in advanced stages, therapy with DBS is indicated. The target region is the globus pallidus internus. DBS is also used in the rare early adult Parkinson’s syndrome with an incidence of 1.5:100,000 (ORPHA:2828), where the target region is usually the Nc. Subthalamicus.
The further goal of the project is the ongoing development and testing of a multi-modal, multi-parameter, platform-independent focused ultrasound system (3MP-FUS) for neuromodulation in dystonia and rare forms of Parkinson’s disease. 3MP-FUS will be integrated into different MRI and PET/MRI platforms for precise targeting of circumscribed brain regions and altering their function.
The approach proposed here has the potential to significantly improve neuromodulation. For brain research and novel therapy, the 3MP-FUS device will open up applications similar to and beyond TMS.
Sponsors: Federal Ministry for Economic Affairs and Energy (BMWi),
Im BMWK geförderten Projekt KliNet5G wird die Umsetzbarkeit einer rein 5G-basierten Netzinfrastruktur auf Basis von OpenRAN in Kliniken evaluiert. Das Projekt verbindet Enduser-Equipment-Hersteller, Klinikbetreiber und medizinische Anwender. Es werden unter anderem Konzepte für die zukünftige Ausgestaltung der Infrastruktur und die damit einhergehenden Veränderungen von Arbeitsabläufe entwickelt. Außerdem werden praxisnahe klinische Anwendungen der Logistik und Patientenversorgung kombiniert und umgesetzt, um damit Prozesse in der Klinik zu flexibilisieren und kontinuierlich zu optimieren. So kann z.B. mobiles Patientenmonitoring sowie Tracking von Geräten und Equipment praktisch realisiert werden. Das Projekt zielt darauf ab, vorhandenes 5G-Knowhow und 5G-Technologie in vorhandene Produkte und Anwendungsgebiete der Medizin zu integrieren um die Anwendung dieser Zukunftstechnologie zu unterstützen.
Sponsors: ZIM – Central Innovation Programe for Small and Medium-sized Enterprises,
The research project SDC – Control Station Med (SDC-CSM) aims at integrating the new communication standard IEEE 11073 SDC – which offers an open, safe and multivendor-capability interconnectivity between medical devices – into a novel control station. The control station then allows the personal of a medical-technical department of a clinic to access aggregated data on the current state of all attached SDC systems. Additionally, SDC-CSM shall provide the documentation of error messages, the handling of errors with automatically specified reactions and the survey of performance numbers. The project will review the possibility and integration of predictive maintenance algorithms. The research centers on the expansion and advancement of SDC standards, data model and data aggregation, and machine learning algorithms.
Sponsors: ZIM – Central Innovation Programe for Small and Medium-sized Enterprises,
Tissue perfusion and moisture are important physiological parameters that reflect the healthy state of patients and are therefore measured for patient monitoring. Problems, such as incorrect drug concentration, pulmonary complications and inefficient oxygen therapy, can be early detected based on the parameter values. Currently, the standard methods, such as pulse oximetry and transcutaneous electrodes, have limitations especially for an application to premature babies. The devices are in contact with the body and measure the local perfusion.
The goal of the MultiGuard project is the development of a contactless and non-invasive multispectral system to support the diagnosis of patient complications. Multispectral imaging combines the principles of photometry with digital imaging and does not require any contrast agent. The system includes a multispectral measurement unit and image processing tools to compute continuously perfusion and pulsatile parameters, fat and water content from the measured absorption values. The light source unit will be made of switchable LEDs, not to disturb the patient with continuous visual light. The physiological parameters have to be delivered at video rate and quality. The visualization has to be optimal to warn the medical staff in case of complications.
At the end of the project, a prototype of the developed system will be evaluated at the intensive care unit and neonatology department.
Sponsors: Deutsche Forschungsgemeinschaft (DFG),
Minimally-invasive endoscopic surgery is a well-established surgical practice. However, decoupled hand-eye-coordination, limited field-of-view and operating space as well as decreased depth perception, are demanding for both surgeon and equipment. Faced with this complex intraoperative environment, surgeons are required to train their spatial awareness and instrumentation skill from training and live operations. Since training effects on spatial cognition and orientation capabilities vary individually, the quality of laparoscopic training with physical and virtual simulators is dependent on the predisposition of trainees. The training effectiveness and a potential skill transfer to the operating room is generally not predictable.
As a consequence, the purpose of this project is the development of a novel training assistance systems that acquires a continuous multimodal representation of a trainees’ individual laparoscopic exercises to predict the current and overall training progression and, in response, provide aural and visual feedback cues. A physical simulator extended with multiple sensor components will be used to generate a knowledge base of basic bimanual laparoscopic skills. Training progression and quality, currently assessed through subjective skill questionnaires, will be extended through the introduction of objective, machine-readable metrics as a form of unbiased description of laparoscopic expertise.

Sponsors:
Validation of the EOS digital information system for German civil protection
The Emergency Operating System (EOS) project is investigating the applicability of a digital, resource-saving software solution for mission and treatment documentation for German disaster relief and civil protection organizations. Based on a digital documentation and information system developed for internationally active emergency medical teams and drawing on experience gained from the EU Civil Protection Mechanism, national workflows, organizational requirements, and technical conditions are systematically evaluated. A central focus is on offline usability, so that the system can be operated reliably even without an internet or mobile phone connection under real operational conditions. The aim is to validate EOS as an electronic documentation and management tool for major incidents and to identify necessary adjustments for use in Germany.
This measure is co-financed by tax revenues based on the budget approved by the Saxon State Parliament.

Sponsors: Janssen-Cilag GmbH,
The medical field of hematology is characterized by highly heterogeneous diseases and disease courses. Nevertheless, clinical trial design, drug development and subsequent therapy are mostly based on the administration of identical therapeutic regimens. As treatment strategies become more precisely tailored to patients, this process becomes more effective, but at the same time causes an enormous amount of complexity in the information that must be considered. Thus, clinical decision-making also depends on whether the treating physician has the appropriate therapeutic experience and access to novel therapies. The goal of KAIT, an artificial intelligence-based platform for therapeutic decision support for patients with myelodysplastic syndrome, acute myeloid leukemia and multiple myeloma, is to support the clinical decision-making process by providing and evaluating relevant information to enable patient-specific and personalized treatment for all patients.

Sponsors: Saxony,
Annually more than 270,000 people suffer from acute strokes in Germany, out of which roughly 200,000 experience it for the first time. Further consequences follow after the acute event for the majority of the afflicted patients, including physical handicaps and emotional distress. This often leads to the stroke evolving into a chronic illness.
In order to support patients in the often difficult time after the stroke, the PostStroke Manager represents a patient-oriented, digital system that uses common everyday communication channels in order to enable a coordinated preventive long-term care of stroke patients. The system integrates stroke pilots (Schlaganfalllotsen), patients and general practitioners as well as mobile sensors (so-called wearables) and, thus, creates the basis for innovative digital services, new forms of care and a structured disease management program for strokes. It serves as a supplement to stroke pilot care from the first year on after the acute event, but can also be used in areas without an established program of that kind directly after the incident. In addition, the PostStroke-Manager can help with secondary prophylaxis matters by i.e. reminding the patient to take their medications and – provided that the patient enabled the function – recording important parameters like heart rate or systolic and diastolic blood pressure via Bluetooth blood pressure monitors.

Sponsors: Federal Ministry for Economic Affairs and Energy (BMWi), ZIM – Central Innovation Programe for Small and Medium-sized Enterprises,
The project aims to develop a non-invasive, multimodal monitoring system that will enable first responders to obtain informative objective feedback on the quality of executed cardipulmonary resuscitation. Regardless of their technical or medical background knowledge, users should be able to quickly bring the system into an operational state, attach it to the patient’s body and receive an evaluation of the resuscitation activity performed. The user should be shown the qualitative changes in the arterial blood flow in the neck that occurs as a result of thoracic compressions. In addition, several preclinical measurements on the phantom are to be used to establish ranges for measured values in the laboratory environment, which will support the user in assessing the displayed blood flow values.

Sponsors: Federal Ministry of Education and Research (BMBF),
In the MOMENTUM project, medical technology is being developed that can be used not only in the hospital (eg in the trauma room, operating theater, intensive care unit), but also in preclinical care at the scene of the accident and in the ambulance. … (more informations)

Sponsors: Federal Ministry for Economic Affairs and Energy (BMWi),
Due to the new Medical Device Regulation of the EU, the requirements for reliable medical device data are continuously increasing. To prove their continuous safety and performance, a market observation of the products based on clinical studies is explicitly required, which also enables a comparative quality and performance evaluation. The market monitoring is expected to optimize the performance data of medical devices, but also to improve diagnosis and medical treatment. The acquisition, storage, and analysis of clinical data in compliance with the data protection regulations are essential for this evaluation. Currently, the market surveillance is often characterized by a lack of skilled personnel in industry and hospitals, cost pressure, legal uncertainty and IT systems with low interoperability.
These aspects will be addressed in the AIQNET project (Artificial Intelligence for Clinical Studies). With the help of artificial intelligence epidemiological, clinical, para-clinical and radiological data can be automatically analyzed and used for the performance evaluation of medical devices.
Therefore, a digital ecosystem that ensures compliance with legal and ethical frameworks through state-of-the-art architecture and security technologies will be conceptualized and developed in the project. Within the framework of the platform, a database will be created, which enables both the clinics and medical device manufacturers to use clinical data for research and development effectively and in compliance with the EU regulatory framework.
As part of the sub-project, ICCAS will develop a “Digital Patient Model” that integrates different perspectives on treatment and the patient. The aim is to improve AI-supported clinical quality and performance assessment based on previous knowledge about the patient and the treatment. This includes aspects of diagnosis and the disease, information on therapies and the specific characteristics of the patient.
Sponsors: KARL STORZ SE & Co. KG,
Aim of this project is to develop a real time pulse oximetry system based on endoscopic multispectral imaging. Relevant medical applications will be determined and preclinically evaluated.
Sponsors: KARL STORZ SE & Co. KG,
The project aims to acquire and analyse hyperspectral data with a laparoscopic HSI-system in the clinical context. Furthermore, methods for the automated classification and visualisation of tissue with hyperspectral data will be developed and implemented.
Sponsors: Federal Ministry of Education and Research (BMBF),
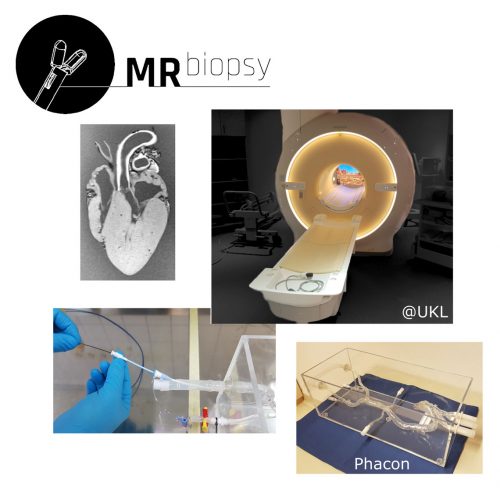
The goal of the joint project ‘MR-Biopsy’ is to research a worldwide first MR safe flexible biopsy forceps. This should improve the minimally invasive and image-guided removal of tissue samples without the previous high X-ray and contrast agent exposure. … (more informations)
Sponsors: Federal Ministry of Education and Research (BMBF),
Das Projekt SONORAY hat zum Ziel, therapeutischen fokussierten Ultraschall (FUS) und Strahlentherapie (RT) zur Behandlung von malignen Tumoren sowie Tumormetastasen zu kombinieren. Die zugrunde liegende Hypothese ist, dass zwei Gewebe zerstörende Methoden/ Energien (Energie von hochintensiven akustischen Wellen und ionisierender Strahlung) effektiver im Kampf gegen Krebs sind, als wenn jeweils nur eine der beiden Energien angewendet wird. … (more informations)

Sponsors:
Image-guided medical interventions gain more and more clinical acceptance. The imaging via ultrasound, X-ray computed tomography or magnetic resonance tomography allows minimal- and non-invasive procedures such as biopsies, thermal ablation, and embolization. Image guidance leads to smaller incisions and thus reduces inflammation and hospitalization. … (more informations)

Sponsors:
In cooperation with the Max Planck Institute for Cognitive and Neurosciences and the Fraunhofer Institute for Biomedical Engineering (IBMT) in St. Ingbert, the possibilities of the application of Focused Ultrasound (FUS) for neuromodulation and neurostimulation are investigated. … (more informations)
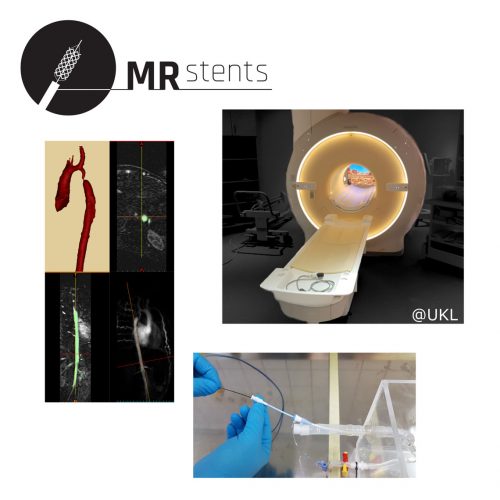
Sponsors: Federal Ministry of Education and Research (BMBF),
Goal of the joint project MR-Stents is the development of a Magnetic Resonance (MR)-guided stent implantation. MR imaging obviates the exposure to radiation and contrast media and enables an increased differentiation of the heart, blood flow analysis, and a 3D display of the heart – enhancing the stent implantation procedure. … (more informations)
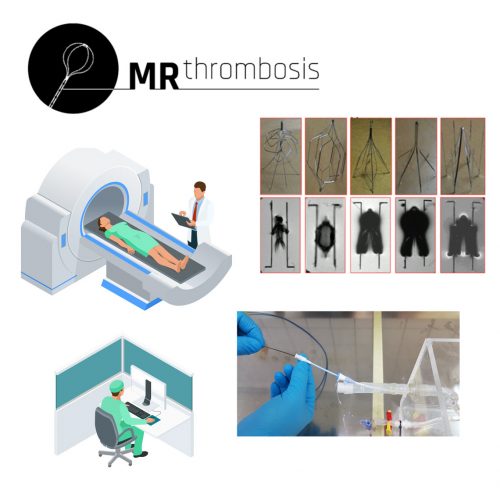
Sponsors: Federal Ministry of Education and Research (BMBF),
The goal of the joint project MR-Thrombosis-Theranostics is the research and development of a Magnetic Resonance (MR)–guided diagnosis and minimally-invasive therapy of thromboses. MR-guidance overcomes the aforementioned disadvantages of X-Ray fluoroscopy and adds high soft tissue contrast. … (more informations)
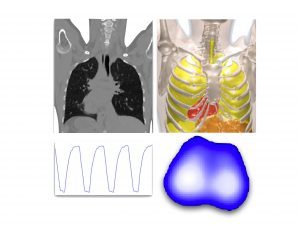
Sponsors: Federal Ministry for Economic Affairs and Energy (BMWi),
A multi-modal ventilation system combined with an EIT measurement device will be developed to be used for temporary monitoring and automated ventilation of the patient. Vital parameters such as lung activity and lung volume will be measured. Additionally, a thorax belt integrates EIT-based imaging of the lung. … (more informations)
Sponsors: ZIM – Central Innovation Programe for Small and Medium-sized Enterprises, Federal Ministry for Economic Affairs and Energy (BMWi),
Project description
Thermography measures the electromagnetic radiations emitted by a warm object which temperature is above absolute zero. These invisible radiations for the eyes are converted by the sensors in the infrared camera into images, whose pixel intensities are proportional to the measured temperatures. This imaging modality is non-invasive, contactless and easy to use. The field of applications of thermography in the medical area is bright: monitoring of vascular and rheumatic disorders, detection of breast cancer or study of tissue vascularization. Since the acquisition of images requires no contact with the patient, this technique is optimal for an intraoperative use.
Nowadays, a large set of infrared cameras are available on the market, but not any model is optimal for the operating room. Our requirements are: a good optical and thermal resolution, the detection of minimal temperature differences, the compatibility of the system with the surgical environment (volume of the device, mobility, facility of use). Moreover, the acquisition, visualization and analysis of the thermograms (or infrared images) are performed by software providing general and basic methods. More sophisticated tools need to be developed for the interpretation of the thermograms, especially in the case of dynamic thermography.
Therefore, the development of a new DIRTH-system (Dynamic InfraRed THermography imaging system) should facilitate the use of thermography in the operating room and improve the interpretation and analysis of thermograms.
Research goals
Goal of this project is the development of an optimal infrared thermography imaging system suitable for the operating room. The system has to be light, compact and transportable, so that it takes little place in the operating room and can be easily moved from place to place. Tools for an optimal visualization of the image data and an optimal analysis of static and dynamic thermography are implemented. The evaluation of the DIRTH-system and its functions is performed with our clinical partners of the Leipzig University Hospital in the operating room on selected applications in the area of plastic surgery.
Project priorities
•Development of a new compact and transportable system for dynamic thermography for intraoperative use
•Development of new functions for the analysis of dynamic thermography
•Evaluation in the operating room in the area of plastic surgery
Sponsors: Federal Ministry for Economic Affairs and Energy (BMWi), ZIM – Central Innovation Programe for Small and Medium-sized Enterprises,
The multidisciplinary treatment of patients in head-and-neck tumor therapy is a challenging problem for the clinicians involved due to the huge amount of available information. This information stems from diagnostic methods such as patient history, blood count and biopsy results as well as medical imaging techniques like computed tomography (CT), magnetic resonance imaging (MRI) and positron emission tomography (PET). In order to support the clinicians, these complex information entities must be processed in a meaningful way. The project OncoControl is the realization of a web-based system which is intended to support the head and neck oncology in exploration, analysis and management of patient data.
The central idea in developing OncoControl is the improvement of the oncological therapy decision-making processes in the field of complex head and neck tumors, improving existing work and treatment processes as well as a support of the decision-making process in the tumor board through the use of endoscopy images and 3D tumor reconstructions alongside the current unused slice images from computed tomography (CT) and magnetic resonance imaging (MRI).
Additionally, a new and innovative form of presentation, the “treatment summary”, supports the clinicians after opening the patient file with a compressed representation of all relevant information in the current treatment step and provides a quick overview of the current patient status.
Associated publications:
Birnbaum, Zebralla, Boehm, Dietz, Neumuth. „Metric Learning for TNM-Classifications of Patients with Head and Neck Tumors“. CARS 2016 Proceedings. Heidelberg, 2016.
Meier, Jens, Stefan Bohn, Bernhard Glaser, Klemens Birnbaum, Andreas Boehm, und Thomas Neumuth. „The Treatment Planning Unit: Concept and realization of an integrated multimedia decision support system for multidisciplinary team meetings“. In MedInfo 2015. Sao Paolo, 2015.
Meier, Jens, Andreas Dietz, Andreas Boehm, and Thomas Neumuth. “Predicting Treatment Process Steps from Events.” Journal of Biomedical Informatics 53 (February 2015): 308–19. doi:10.1016/j.jbi.2014.12.003.
The development of the clinical information system OncoControl allows the physician to get all information about existing patients in a consistent and structured way as well as to retrieve clinical documents such as medical reports or surgery reports. Furthermore, OncoControl allows the clinicians to create and make use of existing information for clinical trials, quality management or conducting clinical certifications. Building on the established central database innovative and knowledge-based methods will assist the clinicians, for example for the TNM classification or document generation in diagnosis, treatment strategy, documentation, as well as presentation and patient education and thus facilitate their clinical practice and the communication between professionals. The use of automated procedures should also pave the way for standardized procedures and comparable analysis results.

Sponsors: ZIM – Central Innovation Programe for Small and Medium-sized Enterprises, Federal Ministry for Economic Affairs and Energy (BMWi),
Eventor is a project in cooperation with the SWAN – Scientific Workflow Analysis GmbH. The aim of the project is the development of concepts for interconnecting process logic and integration technologies for intra- and peri-operative purposes. This allows the semi automation of data exchange and interaction between medical devices based on the surgical situation. Hence, novel forms of surgical assistance might be developed. Additionally, human machine interaction might be simplified to unburden the surgeon and the OR-staff. The implementation is mostly based upon existing integration technologies as well as a tailored rule set for handling of process information. The developed concepts and prototypical implementations will be evaluated for selected clinical fields of application.
The increasing number of medical devices and information systems around the operating theatre lead to additional administrative workload. The project evaluates the application of process information to simplify these tasks. This includes the following research goals:
Eventor focusses on three research topics:
Currently, we analyzed clinical fields of application and their requirements towards dynamic integration of process logic. Furthermore, existing technologies in various fields of research had been evaluated.
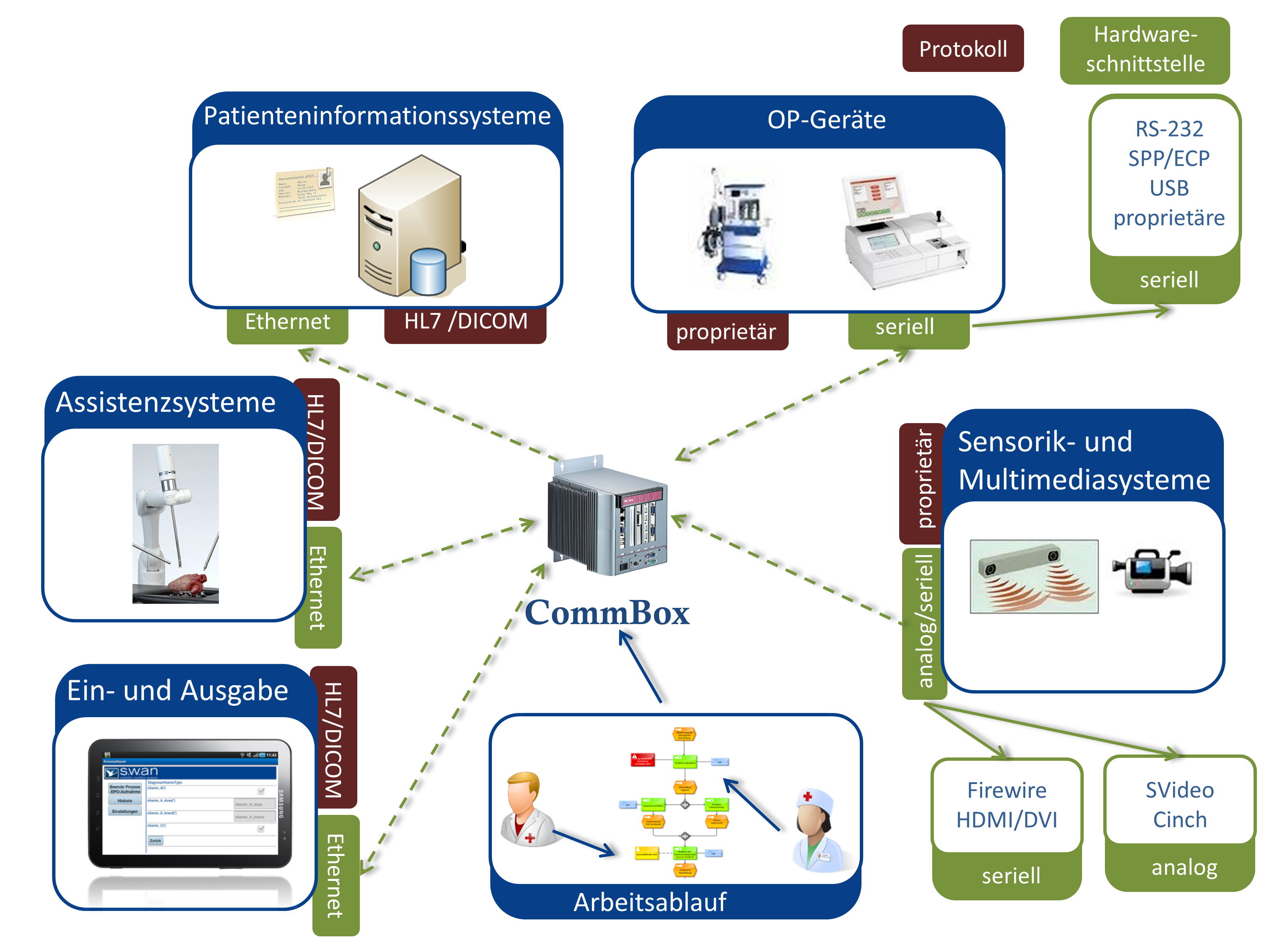
Eventor develops and evaluates concepts for dynamic interconnection of process logic and integration technologies for various clinical applications. The developed concepts and prototypes will contribute to the development of situation-aware medical devices.
Sponsors: Saxony, EFRE,
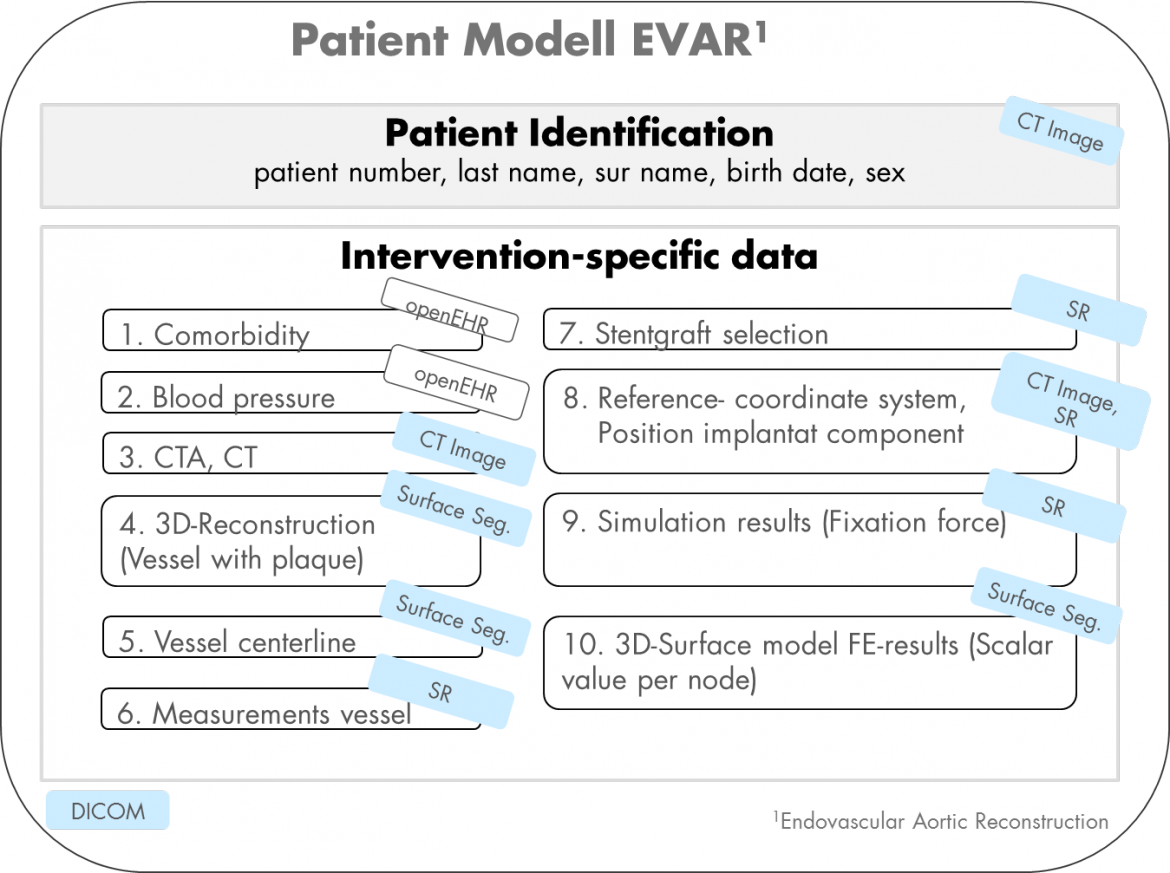
The project deals with the definition of requirements for surgical planning software, which will focus on the surgical use of stent grafts. Furthermore the feasibility of a physically based simulation model for the assessment of stent graft fixation and sealing potential are studied and requirements for a software module for analyzing the results in the medical environment will get defined. The writability of the resulting data is based primarily on existing standards. Required extensions are also included.
A common complication of endovascular aortic aneurysm exclusion is the slipping of the stent graft resulting in insufficient sealing of the vessel. This post-operative complication may be associated with the selection of the vascular implant in the compound. In particular the determination of the stent graft oversize for the fixation of the implant in the vessel is of crucial importance.
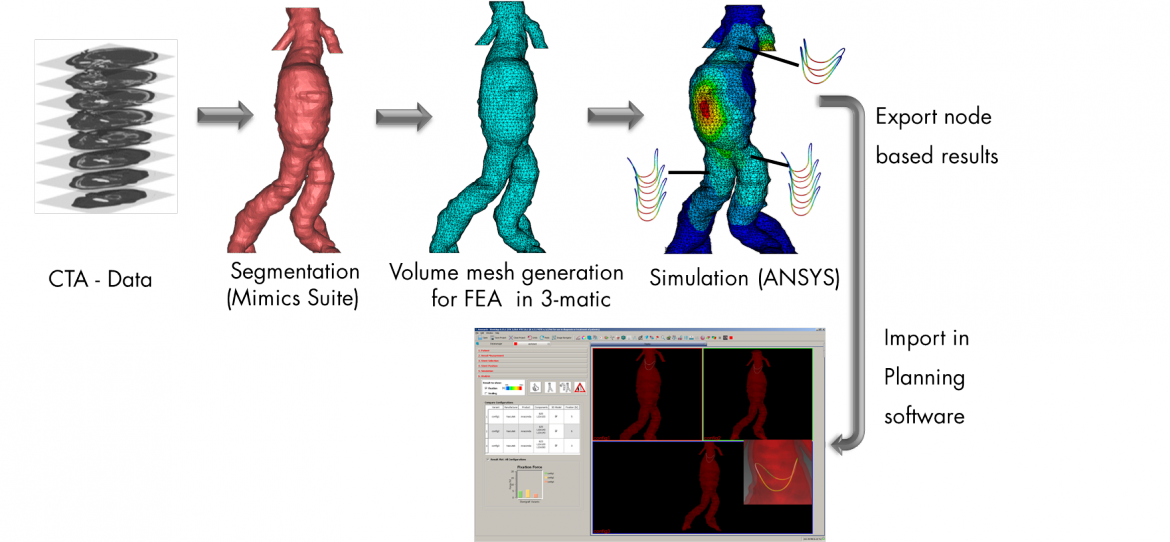
Figure 1: Integration of Calculation Results (FEM Model) into Medical Planning Software for the Evaluation of Stent Graft Properties (Fixation and Sealing Potential)